|
Variation Of Oxidation State Along a Period. By going through the periodic table, we see that the electron-dot symbol of atoms will never have more than eight dots around the atomic symbol. Periodic Trends in the Oxidation States of Elements. It is the transition chemical element with a high atomic value that has variable valency. Lead has two valencies which may be either of 2 or 4. You can understand the interaction of valence electrons with this dot diagram in a better way. With the next element, sodium, the process starts over with a single electron because sodium has a single electron in its highest-numbered shell, the n = 3 shell. Counting valence electrons for main group elements (Opens a modal) The periodic table - transition metals (Opens a modal) Practice. The diagram depicts the numbers of valence electrons of atoms around the symbol of lead. Five 3 d orbitals are filled by the next 10 elements, the transition metals, followed by three 4 p orbitals.\] Consequently, the electron configuration of potassium, which begins the fourth period, is 4 s 1, and the configuration of calcium is 4 s 2. Atoms have a tendency to have eight electrons in their valence shell. The number of valence electrons depends on the octet rule. Elements within the same group share the same number of valence electrons. Atoms in a period have the same number of electron shells. 
Generally, elements in Groups 1, 2, and 13 to 17 tend to react to form a closed shell, corresponding to the electron configuration s2p6. An element period is a horizontal row on the periodic table. Using Electronegativity to Identify Ionic, Covalent, and Polar Covalent Compounds. Electronegativity decreases down a column of the periodic table. That is why elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table. Electronegativity increases in a regular fashion from left to right across a row of the periodic table. Learn how to find the number of valence electrons for main group and transition metals, and how they differ from oxidation state. The number of valence electrons indicates the maximum number of chemical bonds an atom can form. Thus, the periodic table becomes a tool for remembering the charges on many ions. The valence electrons are the electrons in the outermost electron shell of an atom. A valence electron is an outer shell electron that can participate in a chemical bond with another atom. 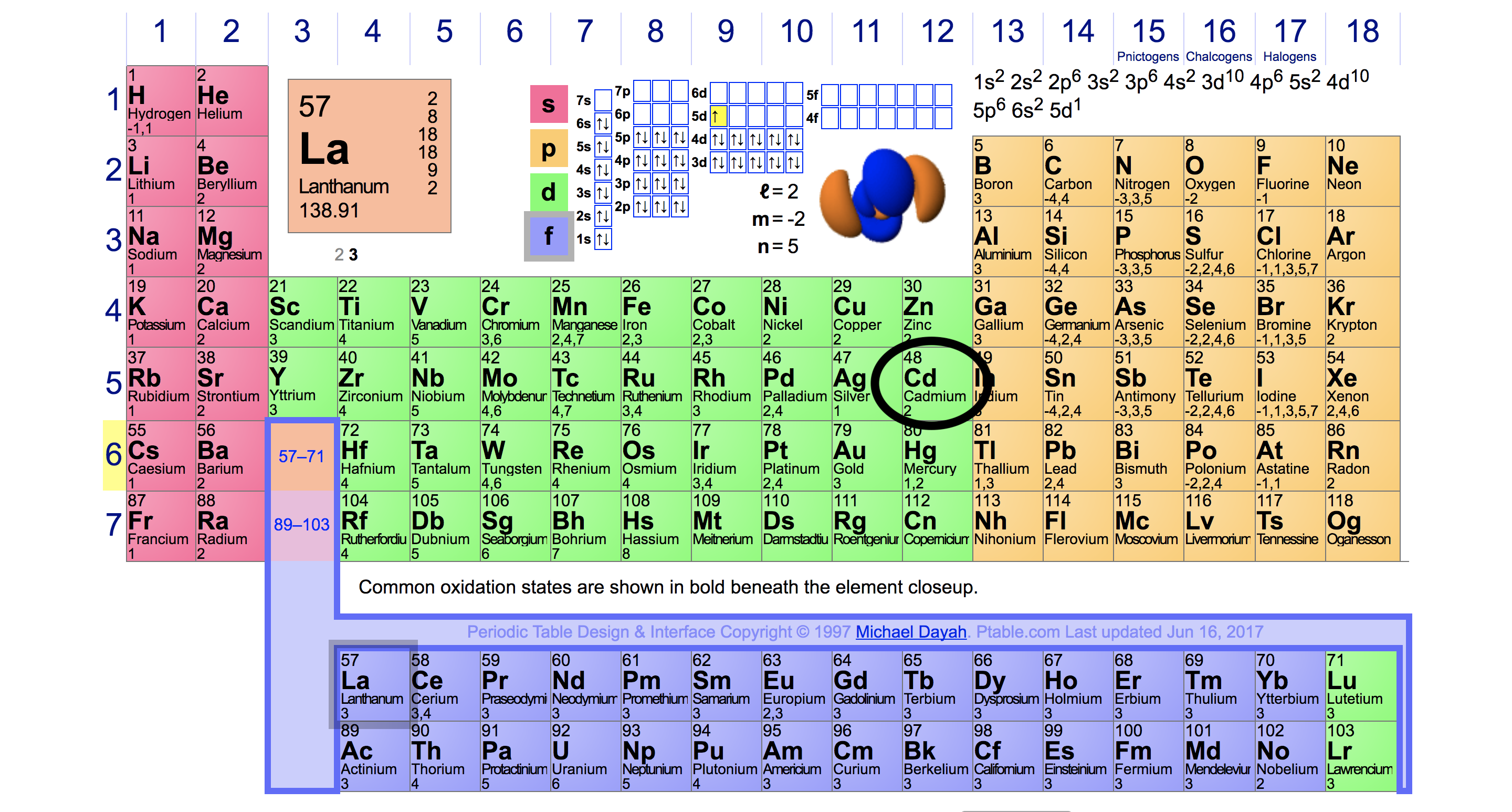
Valence electrons determine the reactivity of an atom. In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number of valence electrons. Accordingly, the 4 s orbital is filled prior to the 3 d orbital because of shielding and penetration effects. Valence electrons are the outer-shell electrons of an atom. Atoms share electrons and form covalent bonds to satisfy the octet rule. This page titled 5.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by Anonymous. The atoms in group 6A make two covalent bonds. Learn what valence electrons are and how to find them on the periodic table. In a similar way, the diagram also provides the presentation of tellurium chemical bonding. Covalent bonds are formed by two atoms sharing electrons. It shows the numbers of tellurium valence electrons of atoms. 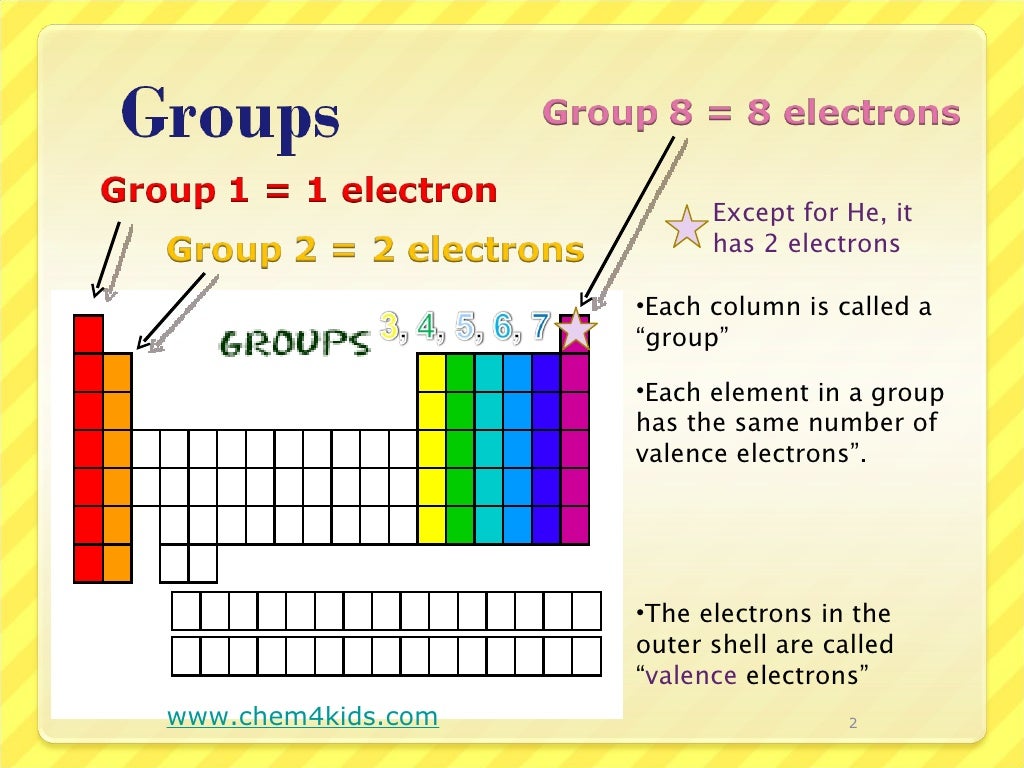
The order in which the orbitals are filled is indicated by the diagonal lines running from the upper right to the lower left. The dot diagram helps in the better representation of Te valence electrons. Subshells corresponding to each value of n are written from left to right on successive horizontal lines, where each row represents a row in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
